-
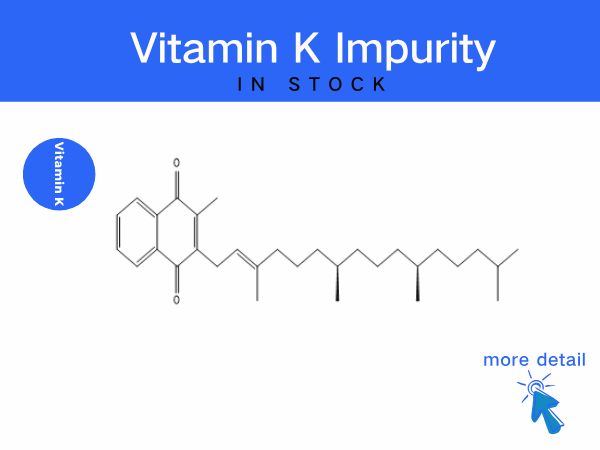
Vitamin K impurity
To address pain points in procurement, SZEB offer a full life-cycle management program: We supplies a complete range of Vitamin K impurities, including:Vitamin K1 Impurity Series...
See More
-
Finerenone impurity
SZEB supplies a comprehensive portfolio of Finerenone impurity reference standards, including stereoisomers such as: (S)-Finerenone、(R)-Finerenone、Finerenone racemate,Below i...
See More
-
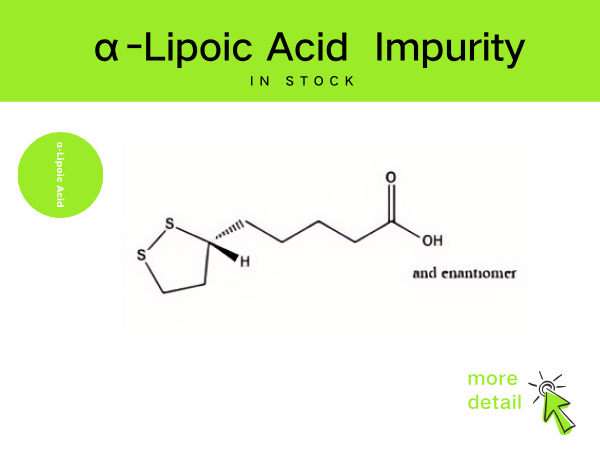
Lipoic Acid Impurity
SZEB supplies a full range of lipoic acid impurities in stock, including the main process impurities in the preparation of lipoic acid, such as: cyclization by-product impurity A , CAS: 12042...
See More
-
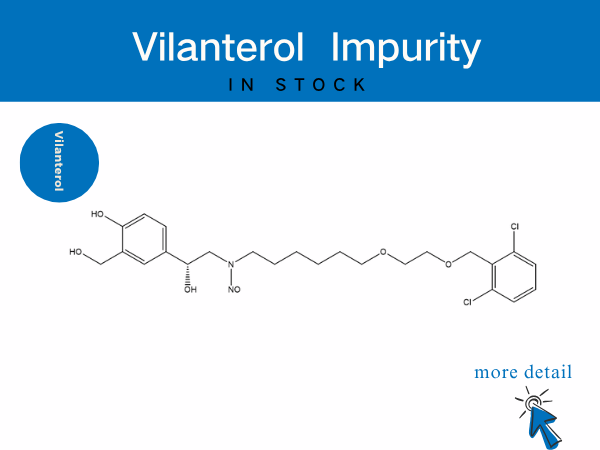
Vilanterol Impurity——in stock
SZEB provides a full suite of vilanterol impurities to accelerate generic drug bioequivalence studies and originator process optimization. Our offerings include: N-Nitroso vilanterol ...
See More
-
Eltrombopag Impurity
SZEB specializes in supplying a comprehensive range of eltrombopag impurities, including:EP-specified impurities、Eltrombopag olamine、Dimer-related impurities These referenc...
See More
-
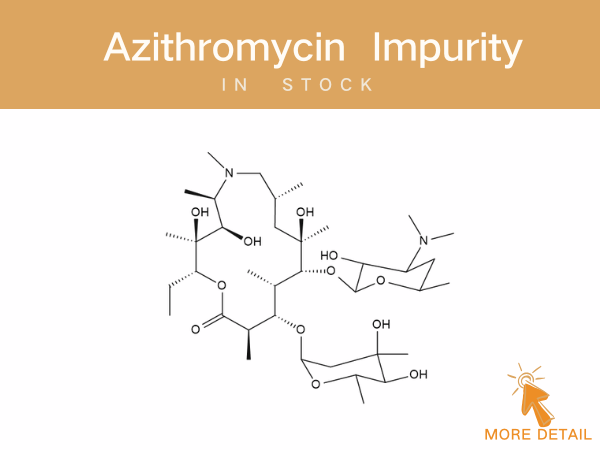
Azithromycin Impurities——in stock
Azithromycin A 11,12-cyclic borate 3’-N-Demethyl-3’-N-(phenylsulfonyl) azithromycin 3-Hydroxy azithromycin acid For detailed impurity profiles and inquiries, visit http://www.ex-biotech.c...
See More
-
Brexpiprazole: A Third-Generation Antipsychotic...
在其实际生产工艺中,会涉及多种布瑞哌唑杂质以及相关研究分析,目前已知部分杂质包括:布瑞哌唑杂质1--50,布瑞哌唑亚砜,结构警示杂质布瑞哌唑氮氧化物等。
See More
-
Abemaciclib impurities
Abemaciclib, the third approved CDK4/6 inhibitor worldwide, was developed by Eli Lilly and Co. It has a broad indication covering early adjuvant therapy (32% reduction in risk of recurrence) ...
See More
-
Salbutamol Impurities
Impurity control is critical to ensuring Salbutamol‘s clinical safety. Impurities may originate from synthesis processes, storage degradation, or packaging materials, and their potential t...
See More
-
Vancomycin Impurity
SZEB specializes in supplying comprehensive vancomycin impurities compliant with EP/USP standards, accompanied by COA, NMR spectra, and traceability documentation. Our services include...
See More
-
Clarithromycin Impurities
Clarithromycin, the star drug in the macrolide antibiotic class, has quickly become a go-to choice in the field of anti-infectives since its successful development by Taisho in Japan back in 1984....
See More
-
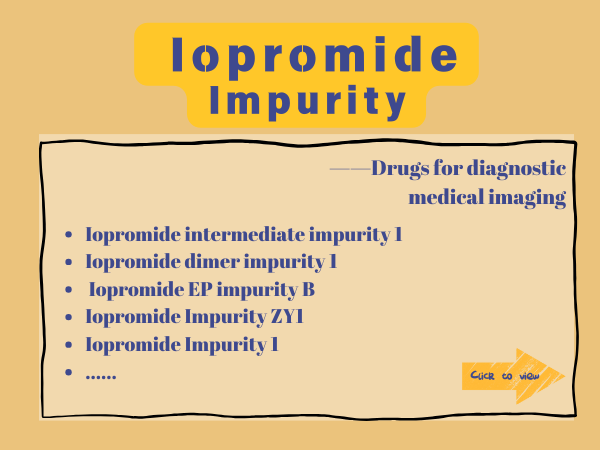
Iopromide impurities
SZEB provides EP/USP certified impurities , isomers, degradation products, etc., providing complete COA, MS/NMR profiles to meet the requirements of FDA/EMA filing. Visit the ...
See More
-
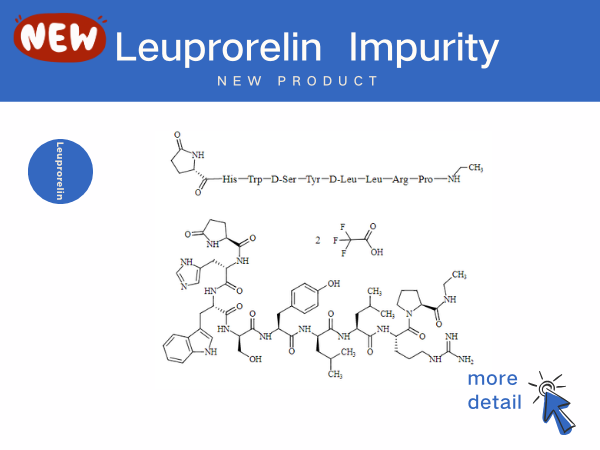
Leuprorelin impurity
In 2025, Changchun Jinsai Pharmaceutical cooperated with Yida Bio to launch leuprolide injectable emulsion (Camcevi) in China, which is a 6-month long-acting, prefilled subcutaneous formulatio...
See More
-
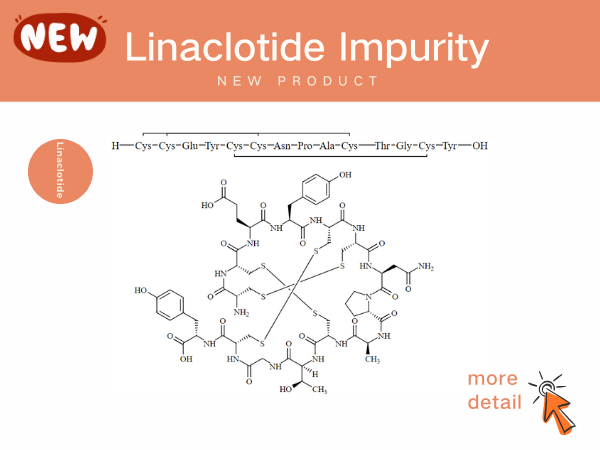
Solution of Linaclotide impurities
As the competition for generic linaclotide has heated up,controling impurity has become the core link for companies to break through technical barriers and capture the market. ...
See More
-
Contrast Agent Medication I: Iopromide
Iopromide is a tri-iodinated, non-ionic, water-soluble X-ray contrast agent widely used in the field of medical imaging diagnostics. It enhances the imaging effects of devices like X-rays and CT...
See More
-
Cefcapene Impurity
Cefcapene, as a third-generation cephalosporin antibiotic, has become an important research target in the field of anti-infectives due to its unique chemical structure and broad-spectrum antibacteria...
See More
-
Cefprozil impurity
SZEB specialize in providing Cefprozil impurities, degradation products and related standards, strictly following USP/EP standards, with purity ≥98%, accompanied by complete COA certific...
See More
-
Ampcilline Impurities
Impurities in ampicillin are of great concern during the medication process, as the presence of impurities may affect the safety and efficacy of the drug. Studies have shown that there are differenc...
See More
-
Lanreotide Impurities
Lanreotide can be used in biological studies concerning the statistical class structure alerts of furans, phenols, nitrosoaromatics and thiobenzenes, and can be used in peptide synthesis....
See More
-
Full set of Cefotaxime impurities in stock...
Full set of Cefotaxime impurities in stock, to meet different experimental needs。Email:sales@exbiotech.com...
See More

















.jpg) Wechat
Wechat